ThermoC: examples
The figure below is a high-pressure phase diagram of the system {ethene + naphthalene}. Naphthalene melts at about 350 K, hence there are solid–vapour equilibria below and liquid–vapour equilibria above this temperature. The compressed ethene acts as a solvent for naphthalene. There are regions in the phase diagram where ethene and naphthalene are completely miscible.
All curves in the figure were calculated with ThermoC, using the Deiters equation of state and density-dependent 1-fluid mixing rules:
- the vapour pressure curve of ethene (component #1)
- the sublimation pressure curve, the vapour pressure curve, and the melting pressure curve of naphthalene (component #2)
- the binary critical curve of the system
- the solid–liquid–vapour three-phase curve originating at the triple point of naphthalene
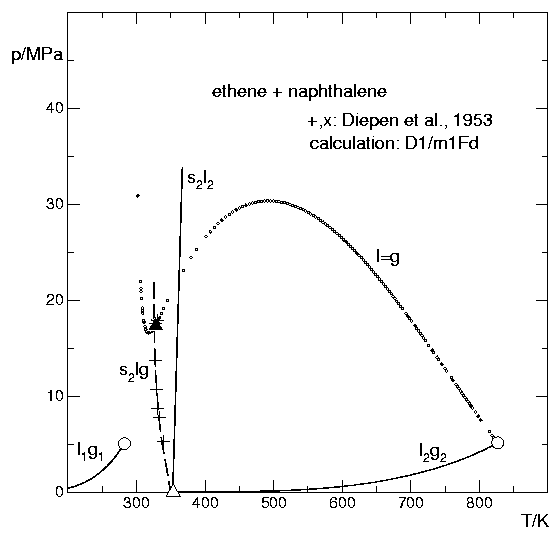
The crosses mark experimental data. All calculated curves were obtained with the same set of parameters.